A Synthetic Biology Approach To Expand Antibiotic Chemical Diversity
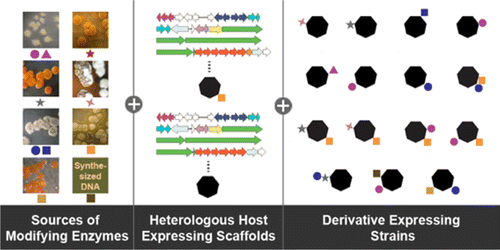
Modification of natural product backbones is a proven strategy for the development of clinically useful antibiotics. Such modifications have traditionally been achieved through medicinal chemistry strategies or via in vitro enzymatic activities. In an orthogonal approach, engineering of biosynthetic pathways using synthetic biology techniques can generate chemical diversity. Here we report the use of a minimal teicoplanin class glycopeptide antibiotic (GPA) scaffold expressed in a production-optimized Streptomyces coelicolor strain to expand GPA chemical diversity.
ACS Infect. Dis., 2016, 2 (9), pp 642–650